Pt100 Resistance Thermometers and Other Temperature Measurement Devices
What Is Temperature?
Temperature is a physical parameter that measures the average kinetic energy of the particles in a system.In common parlance, how warm or cold a substance or place is.
It is one of the seven base units of the SI system.
An increase in temperature means that particles in a system are moving faster and more chaotically, while a decrease in temperature means that particles are moving slower and more orderly.
In order to obtain comparable and reproducible values for temperature, the unit "Kelvin (K)" was defined in the international SI system of units. However, this is usually only used in science or in calculation formulas. In practice, the historically developed units "degrees" are used, which were fixed at certain fixed points of substances (e.g. at "degrees Celsius": melting point/boiling point of water).
Temperature is one of the most important measurands in our daily lives and one of the most commonly measured physical quantities.
Below are some examples:
- Measurement of body temperature in living beings
Indication of diseases etc. - Climate/Weather
With an impact on the growth and reproduction of humans, animals and plants, or even the choice of our clothes and the heating of our homes. - Machines and devices
Must not be overheated or only work optimally at certain temperatures. - Chemical processes and production
Which only take place at certain temperatures - Food
Optimal storage and shelf life of food or preparation of meals.
In most cases, however, an exact determination of the temperature is necessary. This is done by means of various measuring devices, which are also called thermometers in common parlance.
Electronic temperature measuring instruments
Resistance thermometer
Resistance thermometers are temperature sensors that are based on the change in electrical resistance of metals or metal oxides when the temperature changes. The actual sensor element is installed in a so-called protective tube and is thus protected against environmental influences.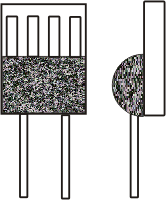
A distinction is made between:- Cold conductor
With temperature changes, some metals already have a sufficient change in resistance at low temperatures
and are therefore called "PTC thermistors". Their resistance increases with an increase in temperature
(PTC = Positive Temperature Coefficient).
Commonly available resistance sensors are made of nickel (Ni100), copper (Cu10), platinum (Pt100, Pt1000) or silicon.
Because of the decisive advantages of sensors made of platinum, these sensors are predominantly used in practice
Pt100 sensors have a nominal resistance of 100 Ohm (Pt100) or 1000 Ohm (Pt1000) at 0°C. - Hot conductor
In addition to temperature sensors made of metal, sensors made of various metal oxides are also used in
mass applications.These often only provide a usable change in resistance at higher temperatures and are
therefore also referred to as hot conductors.
The resistance decreases as the temperature increases (NTC = Negative Nemperature Coefficient). The advantage of these sensors is the inexpensive production and the small design with high response sensitivity.
You can find more information on the other pages of this domain (www.pt100.de)
- Cold conductor
With temperature changes, some metals already have a sufficient change in resistance at low temperatures
and are therefore called "PTC thermistors". Their resistance increases with an increase in temperature
(PTC = Positive Temperature Coefficient).
-
Thermocouples
Pt100-resistance thermometers can be used up to a maximum temperature of 850 °C.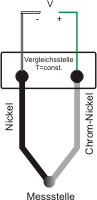
If higher temperatures are to be measured electrically, thermocouples can be used. Thermocouples work according to the "Seebeck effect", named after the discoverer, the German physicist Thomas Johann Seebeck. He discovered that if you weld 2 different metals together and the welded area and the open ends of the wires are at different temperatures, a electrical voltage is created.
This electrical voltage is proportional to the temperature.
As a temperature difference is measured between the welded ends of the wires and the open legs of the wires, the temperature at the reference junction (open legs of the wires) must be known and as constant as possible.
In the past, ice water was used for this purpose. Today, the temperature is measured at the reference junction and the temperature is calculated in the downstream evaluation electronics.
Thermocouples have the advantage that they react very quickly, can measure high temperatures and have a high accuracy.
To protect them from mechanical damage or chemical attack, they are usually installed in protective tubes, just like the Pt100 sensors.
Proven metal wire pairs are standardised and are commercially available as so-called thermo pairs.
Commercially available thermocouples:Please note: The thermocouples Fe-CuNi and Cu-CuNi were available in 2 different alloys. These are not compatible !ThermocoupleTypeMax.temperaturePt30Rh-Pt16RhB1820 °CNiCr-CuNiE1000 °CFe-CuNiJ1200 °CNi-CrNiK1370 °CFe-CuNiL900 °CNiCrSi-NiSiN1300 °CPt13Rh-PtR1760 °CPt10Rh-PtS1600 °CCu-CuNiT400 °CCu-CuNiU600 °C
The types "L" and "U" manufactured according to the old DIN should no longer be used and replaced by the new types "T" and "J".
Mechanical temperature measuring instruments (thermometers)
Liquid-filled thermometers
Liquid-filled thermometers are the oldest type of thermometer used to measure temperature. The measuring principle is based on the expansion of liquids when the temperature changes. A distinction is made between 2 basic designs:- Glass thermometer
Glass thermometers consist of a thin glass capillary tube to which a larger storage vessel is fused at the bottom. The reservoir is filled with a liquid which expands when heated and rises in the capillary tube. When cooling, the liquid contracts and the liquid column in the capillary tube falls.
This expansion is a measure of the temperature.
The measured temperature can be read on a scale printed on the capillary tube.
For better readability, modern capillary tubes are shaped in such a way that a "magnifying glass effect" is created and the thin capillary line is optically magnified. The background is printed white and colourless liquids used (thermometric liquid) are coloured.
As a liquid, almost all liquids can be used within a certain temperature range. They should have as much linear extension as possible.
In practice, mainly mercury, galinstane, ethanol, isopropanol, tuluol, petroleum or pentane are used. The ideal liquid for a wide measuring range from -30 to 350 °C is mercury. The disadvantage of mercury is that toxic vapours are already produced at room temperature, which can lead to health hazards if the glass breaks. For this reason, mercury may no longer be used in the EU since 2009 (except for special applications). Galinstan, a non-toxic alloy of galium, indium and tin that is liquid over a wide temperature range, can be used as a substitute. Galinstan has similar properties to mercury. Galistan, however, has not become widely accepted because of its strong adhesion to glass and the alternative measuring methods that are now available.
In the case of glass breakage with the other thermometric liquids mentioned above, it should also be noted that some of the liquids used are toxic or flammable.
Glass thermometers have no mechanically moving parts like dial thermometers and no electrical energy is required as with Pt100 - resistance thermometers, for example. Therefore, they do not wear out.
Glass thermometers are used as clinical thermometers, in laboratories or with a mechanical protective housing as so-called machine thermometers in the marine sector, HVAC sector and plant engineering.As a more modern variant, there are also machine thermometers as KOMBITEP® in which an additional Pt100 sensor has been installed for remote transmission of the measured values. Typical temperature range: -40°C up to +600 °C
- Dial thermometer with liquid filling
The measuring mechanism of a dial thermometer consists of a storage vessel and a capillary tube made of metal, which are firmly welded to a Bourdon tube mounted in the indicator housing. This measuring mechanism is filled with a thermometric liquid.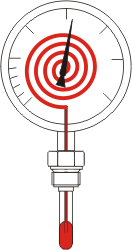
Changes in temperature cause a change in the volume of the thermometric liquid, which leads to a change in pressure due to the closed measuring mechanism.This pressure change is recorded with the Bourdon tube known from pressure measurement technology and transferred to a pointer via a gearwheel system.
The temperature can then be read directly on a calibrated scale. The supply vessel and capillary line are mounted in a protective sleeve that is firmly connected to the indicator housing. By means of an extended capillary line, the protective sleeve (measuring point) and indicator housing can be separated and transmitted over further distances.
It should be noted that the volume of the thermometric liquid in the capillary line is also included in the measurement and leads to measurement errors, especially with long lines.
This can be remedied by factory compensation of the capillary line. Dial thermometers with liquid filling have meanwhile been replaced in many cases by dial thermometers with gas filling. These have some decisive advantages.The increasing temperature measurement with electrical temperature sensors (e.g. Pt100 -rebsistance thermometers) is also pushing this measuring principle out of the market.
- Glass thermometer
Bimetal thermometer
Bimetallic thermometers are mechanical temperature measuring instruments with pointer display.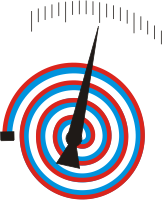
The measuring principle is based on the different expansion of solids when the temperature changes.
The measuring element used is bimetal, which bends when heated. This bending is a measure of the temperature.
Bimetal consists of 2 different, cold-welded metal sheets (e.g. steel and brass) with very different expansion coefficients. The individual sheets are placed on top of each other and rolled at high pressure. By subsequent heating, the two sheets are form-fit joined (cold welded).
When the temperature is increased, the two metals expand differently and the bimetal bends.
In order to obtain the largest possible usable deflection of the bimetallic strip, the longest possible strip is required. In order for this to be used in a space-saving way, the strip is either wound into a spiral or helically. A "bimetallic spiral spring" or a "bimetallic helical spring" is obtained, with which different types of bimetallic thermometers can be built.
Bimetallic thermometers with spiral springs for low temperatures (below 100°C) can be produced at low cost and are therefore often used in the heating, air-conditioning and ventilation sector or as room thermometers.
Temperature range: -70 up to +600°C
-
Gas-filled dial thermometers
The measuring principle is based on the change in pressure of a gas in a closed volume with a change in temperature. The basis is the state formula for gases, according to which the ratio of pressure, volume and temperature of an ideal gas is constant.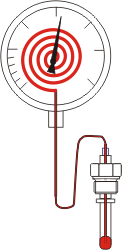
p1 x V1 / T1 = p2 * V2 / T2 = constant.
p = pressure, V= volume in measuring unit (V1 = V2), T= temperature
The measuring mechanism of a gas-filled dial thermometer consists of a storage vessel and a capillary line made of metal, which are firmly welded to a Bourdon tube mounted in the indicator housing. This measuring mechanism is filled with pressurised, non-toxic nitrogen gas.
Since the measuring unit is completely closed, i.e. the volume is constant, a change in temperature leads to a change in the pressure of the filling gas in the measuring unit. This pressure change produces a deflection of the Bourdon tube, which is transmitted to a pointer via a gearwheel system.
The temperature can then be read on a calibrated scale.
The storage vessel and capillary line are mounted in a protective sleeve that is firmly connected to the indicator housing. By means of an extended capillary line, the protective sleeve (measuring point) and the indicator housing can be separated and transmitted over long distances.
The advantage over liquid-filled dial thermometers is that the capillary line has only a very small volume and the volume change of the gas in it can be neglected. Up to 100 m, no compensation line is required to correct the temperature changes in the capillary line.
Measuring ranges: -250 up to + 800 °C
Advantages of gas-filled dial thermometers- Small gas volume in the measuring unit, thus fast response time
- Non-toxic nitrogen gas, instead of toxic mercury or flammable alcohol
(therefore ideal for food applications) - No compensation of the capillary up to 100 m necessary
- No auxiliary energy is required, as is the case with Pt100-resistance thermometers, for example
Non-contact temperature measuring devices / Radiation pyrometers / Infrared thermometers
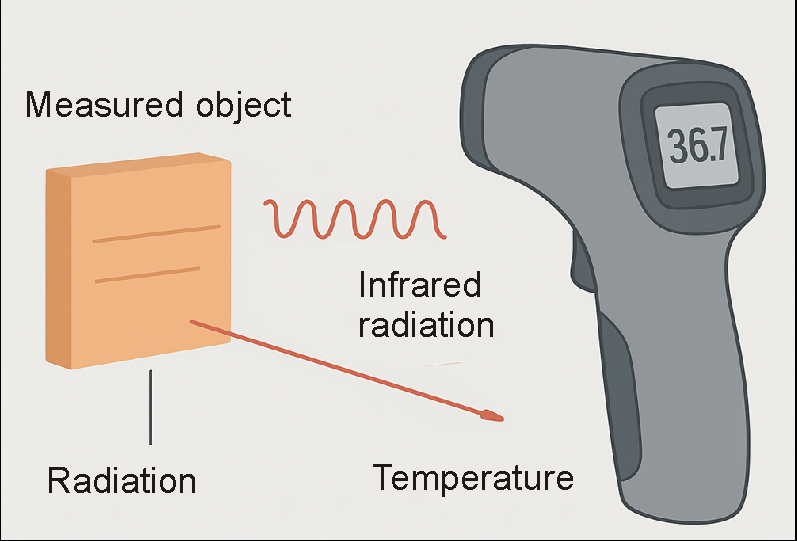
It is based on the physical principle that all bodies with a temperature above absolute zero (-273.15 °C) emit electromagnetic radiation. A large part of this radiation is in the infrared range, which is invisible to the human eye. The intensity and wavelength of this radiation are closely related to the temperature of the surface. The warmer an object is, the stronger and shorter the wavelength of the infrared radiation it emits.
An infrared thermometer makes use of precisely this property: it “captures” the radiation and uses it to calculate the temperature.
For this purpose, an infrared thermometer contains an optical system, often consisting of a lens, which focuses the incoming thermal radiation. This focused radiation hits a detector, usually a thermopile sensor, which converts the thermal radiation directly into electrical voltage and is evaluated by downstream electronics. The more radiation that arrives, the higher the voltage—and the higher the voltage, the higher the measured temperature.
The electronics in the infrared thermometer perform complex signal processing, taking several parameters into account.
For example:
- Emissivity of the material
A decisive factor for precise measurement is the so-called emissivity. This describes how well a surface emits infrared radiation. The value is always between 0 and 1:- 1.0 = perfect black body (maximum radiation emission)
- 0.0 = no radiation emission (theoretical, does not occur in practice)
Infrared thermometers are not very good at measuring bare metals directly because the device often measures the reflection of the surroundings instead of the intrinsic radiation. For bare metals, surface preparation (e.g., adhesive tape, paint, carbon black) or special metal pyrometers are therefore required.
Matte, non-reflective, organic materials are nearly ideal IR emitters (e.g., skin: 0.97–0.98, plastic: 0.93–0.98, matte wall paint: ~0.95, wood: 0.90–0.97). Many infrared thermometers have adjustable emissivity. This allows the user to adjust the device to the material being measured in order to obtain accurate results.
Devices with a fixed emission value are less accurate with certain materials, but remain particularly easy to use. - Ambient temperature (for internal temperature compensation)
- Spectral sensitivity of the sensor
- Optical properties of the measuring optics
- Ambient temperature (for internal temperature compensation)
- Calibration curves of the device
Infrared thermometers are manufactured as compact handheld devices with integrated evaluation electronics and display, or for stationary installation with separate sensor and evaluation electronics.
Measuring spot and distance ratio
With infrared thermometers, the distance-to-spot ratio plays an important role. It describes how large the area is from which the thermometer receives radiation. A ratio of 12:1, for example, means that the device detects a measuring spot with a diameter of 1 cm from a distance of 12 cm.The higher the ratio, the more accurately measurements can be taken from a greater distance. For precise results, always ensure that the measuring spot is completely on the target object.
Advantages of infrared thermometers
- Non-contact
Ideal for hot, dangerous, or moving objects - Fast measurement
results in fractions of a second (making it ideal for food applications) - Hygienic
no contamination, particularly relevant in the food or healthcare sectors - Versatile
works on machines, liquids, walls, or electrical components
Chemical temperature measurement (thermochromic inks, temperature measuring strips)

They are ideal for products where temperature changes need to be made visible—for example, in industry, in laboratories, or in everyday life.
There are two types of thermal inks:
- Reversible thermal inks
These change color at the defined temperature and return to their original color when the temperature returns to normal.
RReversible thermal inks contain microencapsulated dye systems that react to temperature changes. The capsules usually contain a combination of leuco dye, developer, and solvent. When a certain temperature is reached, the chemical structure of the dye changes—the color disappears or appears.
When they cool down again, the dye regains its original structure and thus its original color. - Irreversible thermal inks
Irreversible thermal inks are based on pigments or chemical reactions that change permanently when exposed to heat. If the temperature exceeds a defined threshold, the dye molecules transform in such a way that the color changes permanently. This reaction cannot be reversed, which means that temperature peaks remain reliably visible.
Thermal paints are either applied directly to the object to be measured or affixed as thermal strips. Thermal strips consist of a thin film onto which one or more temperature-sensitive color fields are applied.
If several different thermal paints are applied, a larger temperature range can also be monitored.
Depending on the model, the fields react reversibly or irreversibly.